Environment & Energy
Related: About this forumOperando Monitoring of Plutonium on Ion Exchange Columns Using UV/Vis/NIR Spectroscopy.
The paper to which I'll briefly refer is this one: UV–Vis–NIR Reflectance Spectroscopy and Chemometrics for Monitoring Pu Directly on an Ion Exchange Column Luke R. Sadergaski, Jeffrey D. Einkauf, Hunter B. Andrews, Laetitia H. Delmau, and Jonathan D. Burns Industrial & Engineering Chemistry Research 2026 65 (9), 4673-4680.
I am not dissuaded that the element plutonium, named after a planet that has been declared not to be a planet, is the key to saving this planet. Via conversion to plutonium, we, in theory, could close down every energy mine, including fossil fuel mines, on the planet for centuries, by converting the huge inventory of already mined and isolated 238U to plutonium via the fast neutron spectrum. We could also work to heal the vast destruction of wilderness associated with the so called "renewable energy" scam, and work to restore the land destroyed by these land intensive industrial facilities.
The current industrial procedure for the isolation of plutonium, which is present in used nuclear fuel, depending on "burn-up" (which could be compared to gas mileage on a car) to the extent between 1% - 2%, is the PUREX process, a solvent extraction process which relies on a biphasic mixture of hydrophobic solvents and aqueous solvents. This is not my favorite process; it works fine but is rather problematic in many ways, since the solvents need to be cleaned up, and some of them are petroleum byproducts, kerosene being one example. It is possible to make kerosene from carbon dioxide and nuclear hydrogen using the FT process that Jimmy Carter was fond of while trying to scare OPEC, but my feeling is "Why bother?"
I favor the fluoride volatility process which relies on distillation of volatile metal fluorides which the actinides uranium, neptunium and plutonium form, as do many of the valuable fission products that we need to recover in order to save what is left to save of the world.
Nevertheless, nobody cares what I think, and as result, the aqueous nitric acid solutions resulting from the Purex process are likely be present. Industrial scale ion exchange resins exist, and this paper offers an approach to removing and recovering residual trace plutonium from aqueous nitric acid.
From the paper, out of the Oak Ridge National Lab:
Anion exchange columns are widely used for purifying Pu and rely on the speciation behavior of Pu(IV) in HNO3. (12,13) In dilute HNO3, Pu(IV) predominantly exists as an aquo complex, but as the acid concentration increases, nitrate ligands increasingly coordinate with the Pu(IV) center to form various nitrato complexes. In concentrated HNO3, Pu(IV) forms complexes with nitrate ions to yield anionic species such as Pu(NO3)62–, which interact strongly with the positively charged sites on the resin and allow Pu to be retained while other noncomplexed or positively charged contaminants pass through. (14) The sorbed Pu can subsequently be eluted in dilute HNO3, effectively separating the Pu from fission products and other actinides such as U(VI). (9,15) The efficacy of these anion exchange columns to purify Pu depends on maintaining the proper acid conditions. Techniques such as UV–vis–NIR absorption spectroscopy, which is often combined with multivariate analysis, can detect electronic transitions associated with different Pu(IV) species to provide insights into their relative populations and monitor Pu in process analytical applications. (16−20)
To better understand Pu(IV) speciation on an anion exchange column and improve in situ monitoring capabilities, we introduce a fiber-optic UV–vis–NIR reflectance spectroscopic method that enables direct, in situ measurements on a glass column during Pu(IV) loading and elution. This approach integrates spectroscopic monitoring directly into the column environment to provide continuous insight into chemical changes as they occur. Reflectance spectroscopy measures a material’s absorption properties from the light reflected off its surface; thus, this technique offers a noninvasive method to probe the column interior without disrupting the separation process. (20−23)
In this article, we demonstrate the ability to monitor an ion exchange column directly with a reflection probe and identify unique Pu(IV) species on the resin before elution. The absorption spectra acquired on the anion exchange column are compared with solution analogs over a range of HNO3 concentrations from 0.5 to 13 M...
I'm not going to spend a lot of time going over the statistical principal component analysis (PCA), a statistical means of transforming the vector of multivariate inputs (in this case the three spectral wavelengths) to address the data analysis.
A picture of the apparatus used can be found in the paper, which gives a feel for the apparatus. For the experiments in question, 240Pu was used, which has a fairly high critical mass; the issue of criticality would need to be addressed on a larger scale approach using this technology, but I wouldn't suggest it to be applicable on an industrial scale.
The apparatus in the lab:

The caption:
Because of the many available electronic transitions among the rich array of orbitals available in this heavy f element, plutonium complexes and salts are generally highly colored. The nitrate complex here is green.
Here is the spectra across the range evaluated of the complexes at different acid strength:
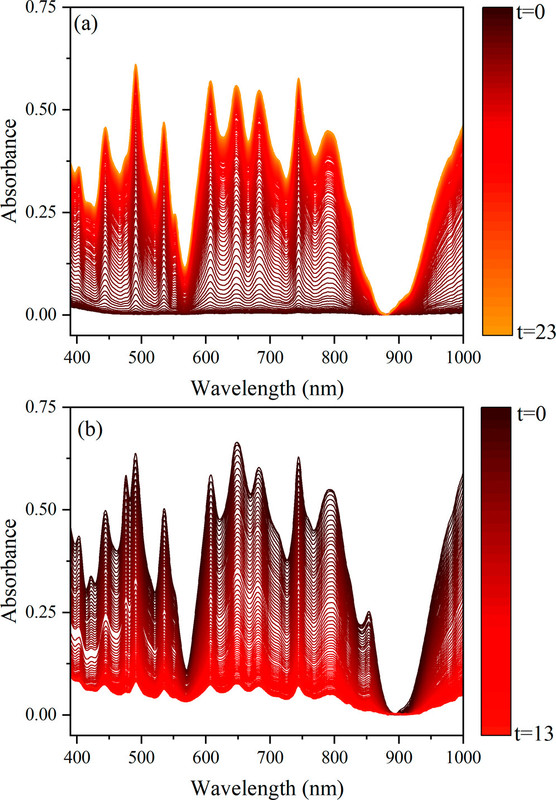
The caption:
From the paper's conclusion:
The ability to probe chemical changes directly on a glass column surface offers significant advantages over traditional effluent-based flow cell approaches. In situ spectral data reveal spatial gradients, resin saturation behavior, and breakthrough dynamics. The fiber-compatible design supports deployment in remote systems, making it well-suited for challenging environments such as radiochemical separations and nuclear process monitoring. In dynamic systems in which the scores trajectory follows a consistent pattern through processing conditions, deviations from the expected trajectory could be used as early indicators of process shifts, making the method useful for real-time monitoring and prediction...
Again, clean plutonium separations are critical to the survival of what is left of the environment, and this paper, while esoteric, is a contributor to making sure that is done cleanly and in a sustainable fashion.
An interesting paper.
Have a nice evening.